Avalanche®-in vivo Transfection Reagent is extremely powerful for gene functional studies and RNA interference in vivo. This reagent was made by our proprietary formula which interacts with nucleic acids, and move into cells via endocytosis. In addition to efficiently transporting nucleic acids into the cells, the formula is also able to lead to the protection of the nucleic acids from lysosomal degradation and the efficient release of those nucleic acids from lysosomes to ensure maximum function. Avalanche®-in vivo has been proven to be able to perform the maximum delivery of DNA, shRNA, siRNA, or oligonucleotides into different organs in the body. No significant pro-inflammatory responses have been detected using this reagent. Avalanche®-in vivo can be administered via various routes, such as intravenous, intraperitoneal, intratumoral injection, etc. Avalanche®-in vivo Transfection Reagent is the best choice for delivering nucleic acids in vivo.
Features:
-
Efficient delivery of DNA, shRNA, siRNA or oligonucleotides in vivo
-
200 µl reagent for 20 transfections in mouse
-
Efficient delivery to the lung, liver, kidney, and spleen, liver, pancreas and certain type of tumor via systemic administration
- Delivery via Multiple modes of administration in many species
- Avalanche®-in vivo/nucleic acid conjugated complexes are stable in serum for 16h
- No detectable inflammatory responses
- Efficient siRNA and plasmid DNA delivery via direct subcutaneous tumor injection (multiple tumor types)
- Reproducible results
- Applicable for plasmid DNA/siRNA co-injection
- Developed and manufactured by EZ Biosystems LLC
Data:
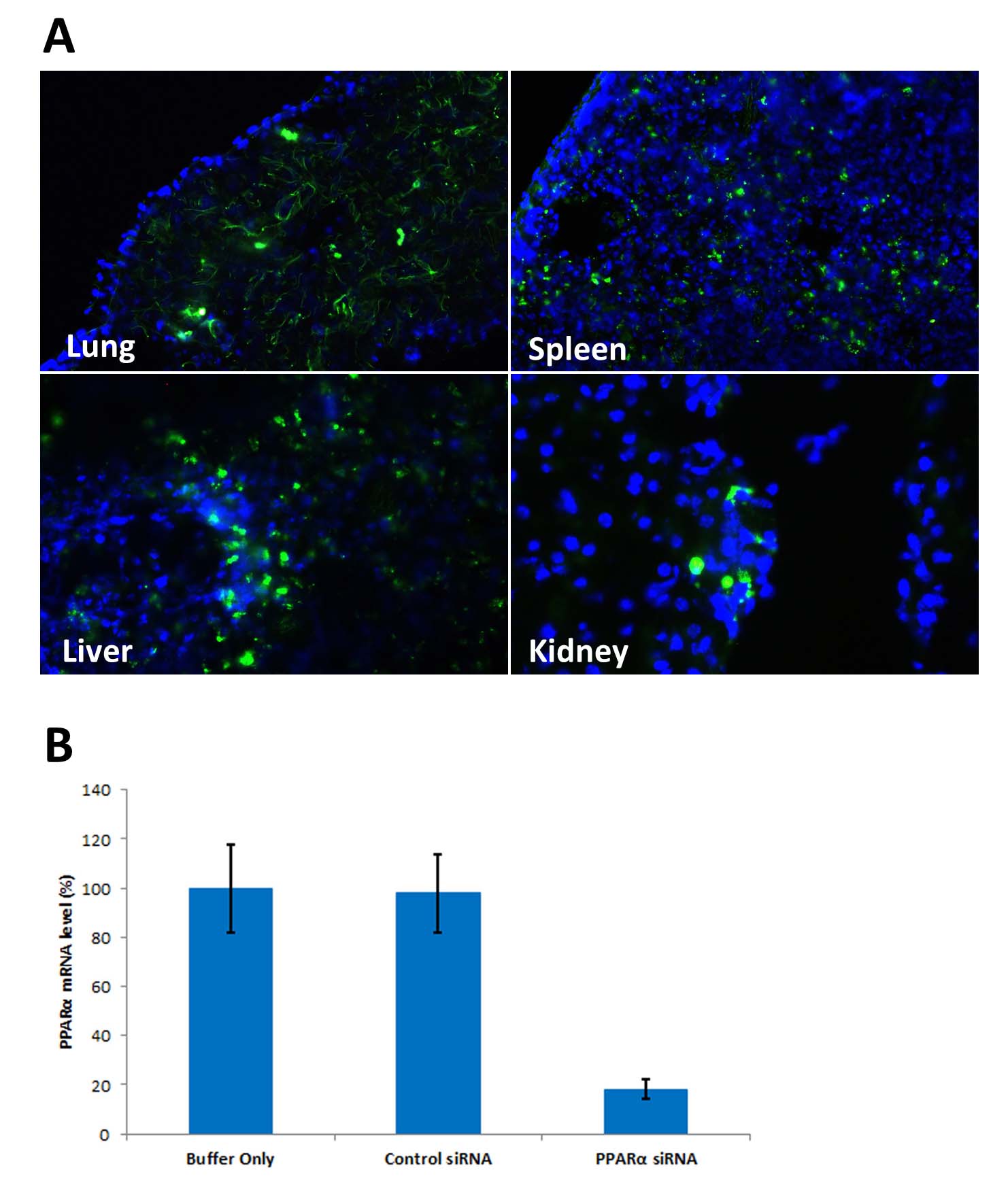 |
| Figure 1. Avalanche®-in vivo Transfection Reagent performs high transfection efficiency in vivo. (A). Avalanche®-in vivo Transfection Reagent successfuly delivered EGFP expression vectors into diffeent organs via systemic administration (tail vein). 48 hours post transfection, different organs were harvested. Frozen sections revealed EGFP positive cells. DAPI was used to stain nucleus. (B)In vivo RNAi test: Systemic administration (i.v.) of Avalanche®-in vivo Transfection Reagent conjugated with siRNA targeting PPARa mRNA or non-silencing control siRNA (30 µg/mouse) following the recommended protocol. Liver was collected and RNA isolated 24 hours after first injection. Samples were analyzed by qRT-PCR for PPARa mRNA expression levels. Ribosomal RNA levels were used to normalize the PPARa mRNA data. Data are means /- SD (n=6). |
| |
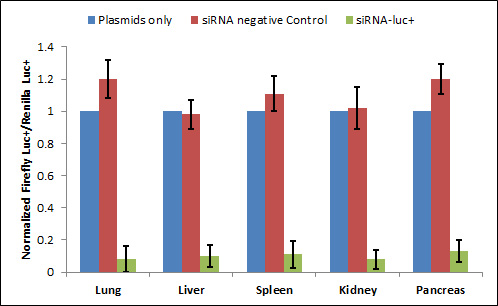 |
| Figure 2. Co-transfection application: RNA interference in organs of mice after transfection of siRNA using Avalanche®-in vivo Transfection Reagent. Mice were co-injected with the pGL3 control vector and pRL-SV40 plasmids (Promega) alone or together with siRNAs. The two plasmids contain genes of P. pyralis luciferase (Pp-luc ) and R. reniformis luciferase (Rr-luc) respectively. In all cases, the ratio of Pp-luc to Rr-luc activities was calculated to compensate for differences in transfection efficiencies between mice. The ratios were then normalized to those observed in mice receiving no siRNA. Blue bars, red bars, and green bars indicate normalized Pp-luc /Rr-luc activity ratios in mice receiving plasmids only, in controls co-injected with siRNA-EGFP, and in those co-injected with siRNA-luc respectively (References method: Lewis et al., Nature Genetics 32: 107-8, 2002, but used Avalanche®-in vivo Transfection Reagent for transfection). |
| |
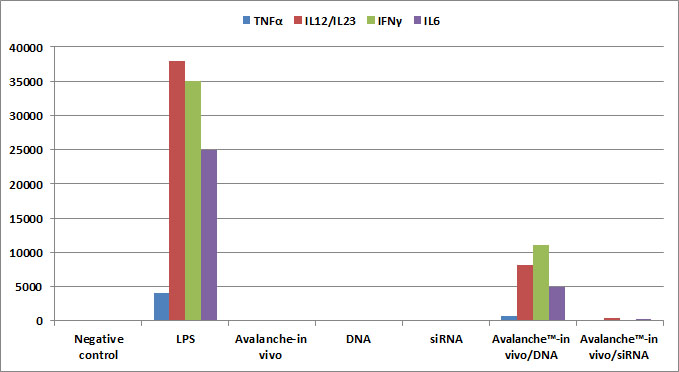 |
|
Figure 3. Avalanche®-in vivo does not cause any significant pro-inflammatory responses: Serum concentration of TNF-a, IL12/IL23, IFNr and IL6 following intravenous nucleic acid delivery using Avalanche®-in vivo Transfection Reagent 6 h after delivery. The negative control consists of an IV injection of 5% glucose; the positive control an IP injection of 50 µg LPS. 40 µg DNA and 40 µg siRNA were delivered with or without Avalanche®-in vivo Transfection Reagent.
|
Protocol
MSDS
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200