5'-GalNAc C3 Phosphoramidite - (10-1974)
(4-(Trimethoxytrityloxymethyl)-1-(6-(4-(3,4,6-O-triacetyl-2-acetylamino-2-deoxy-β-D-galactopyranosyl)butanamido)hexanoyl)piperidin-4-yl)methyl-O-[(2-cyanoethyl)-(N,N-diisopropyl)]
phosphoramidite
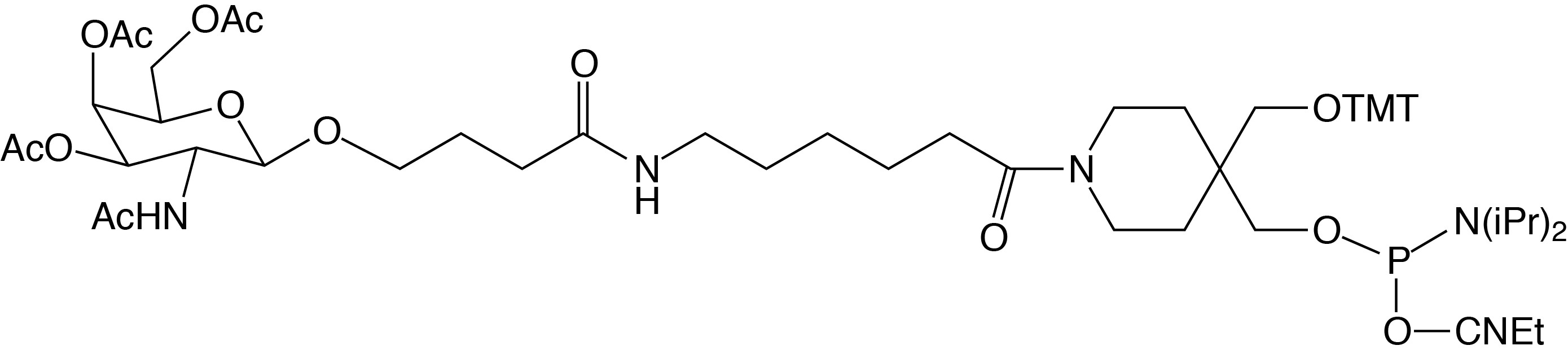
CAS Number: 2437303-51-8
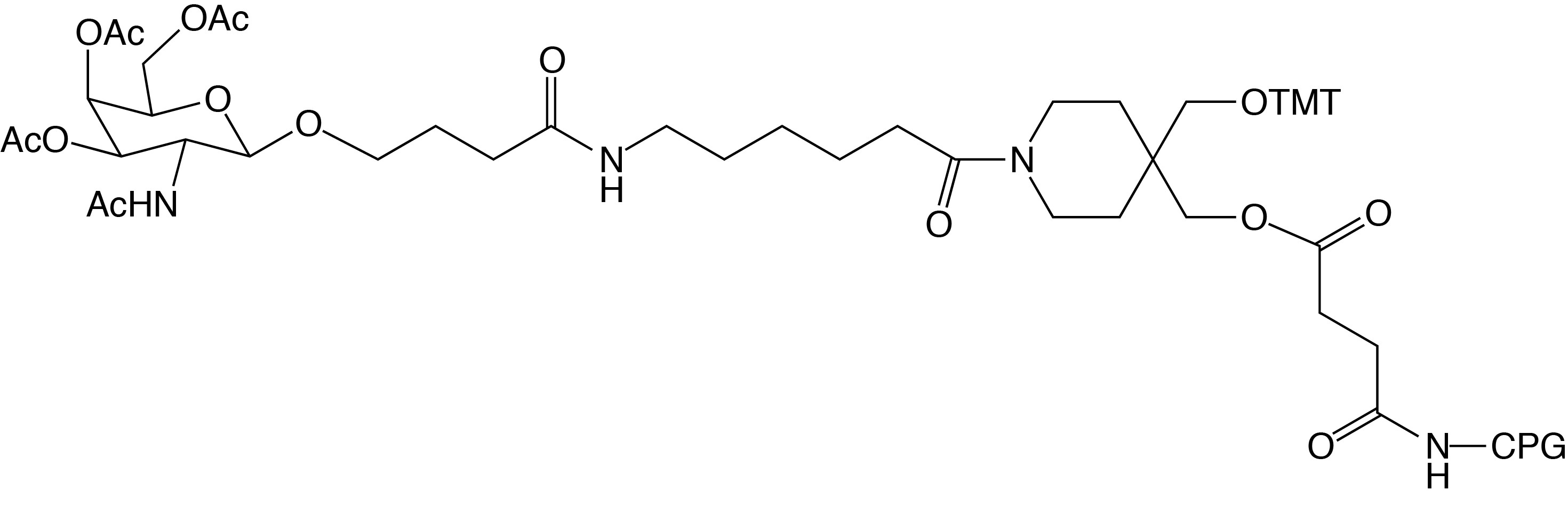
GalNAc C3 CPG - (20-2974)
(4-(Trimethoxytrityloxymethyl)-1-(6-(4-(3,4,6-O-triacetyl-2-acetylamino-2-deoxy-β-D-galactopyranosyl)butanamido)hexanoyl)piperidin-4-yl)methyl-O-succinoyl
CPG
N-Acetylgalactosamine (GalNAc) Oligonucleotide Conjugates
A directed approach to the delivery of therapeutic oligonucleotides specifically to the liver has been to target the asialoglycoprotein receptor (ASGPR) using a suitable glycoconjugate. Indeed, ASGPR is the ideal target for delivery of therapeutic oligonucleotides to the liver since it combines tissue specificity, high expression levels and rapid internalization and turnover. The use of oligonucleotide glycoconjugates has led to significant advances in therapeutic delivery as evidenced by the work of Alnylam Pharmaceuticals and Ionis Pharmaceuticals using multivalent N-acetylgalactosamine (GalNAc) oligonucleotide conjugates.
Glen Research is delighted to introduce a GalNAc modification strategy using a monomeric GalNAc support and the equivalent GalNAc phosphoramidite. Our experimental work has shown that these products are fully compatible with regular oligonucleotide synthesis and deprotection. Oligonucleotides containing GalNAc can be deprotected using standard procedures during which the acetyl protecting groups on the GalNAc group are removed. We have demonstrated that 5'-GalNAc C3 phosphoramidite can be used to prepare oligonucleotides with multiple consecutive GalNAc additions at the 5' terminus.
Glen Research offers these GalNAc C3 products under an agreement with AM Chemicals LLC.
Diluent: Anhydrous Acetonitrile
Coupling: 12 minute coupling time is recommended.
Deprotection: As required by nucleobases. Compatible with 30% ammonium hydroxide for 2 hours at 65°C, 17 hours at room temperature, AMA (ammonium hydroxide/40% methylamine 1:1) for 10 minutes at 65 °C and UltraMild deprotection conditions. Note extended deprotection in ammonium hydroxide (17 hours at 55 °C) will lead to some degradation.
Storage: Refrigerated storage, maximum of 2-8°C, dry
Stability in Solution: 1-2 Days
Please Note: GalNAc C3 products are sold under agreement with AM Chemicals LLC.
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200