ClearColi BL21(DE3) Electrocompetent Cells
Each ClearColi® BL21(DE3) Electrocompetent
Cell Kit contains: ClearColi BL21(DE3) Electrocompetent Cells in DUO
packaging (2 transformations per tube), Expression Recovery Medium
(lactose minus), pUC19 Positive Control Plasmid, and complete
protocols.
Expression Recovery Medium (lactose minus) is also available separately.
Product information
Introduction to ClearColi® technology:
Is there a better way to eliminate endotoxin contamination?
Now there is.
Instead of removing lipopolysaccharide (LPS) contamination from your
protein or plasmid DNA preparations, eliminate the LPS at the source.
Genetically modified LPS from a novel E. coli strain produces
functionally clean recombinant proteins and plasmids. ClearColi®
BL21(DE3) cells are the first commercially available competent cells
with a modified LPS (Lipid IVA - see Fig. 1) that does not
trigger the endotoxic response in mammalian cells. ClearColi™ cells lack
outer membrane agonists for hTLR4/MD-2 activation; therefore,
activation of hTLR4/MD-2 signalling by ClearColi is several orders of
magnitude lower compared with E. coli wild-type cells, and
heterologous proteins prepared from ClearColi are virtually free of
endotoxic activity. After minimal purification from ClearColi cells,
proteins or plasmids, which may still contain Lipid IVA, can still be used in most applications without eliciting an endotoxic response .
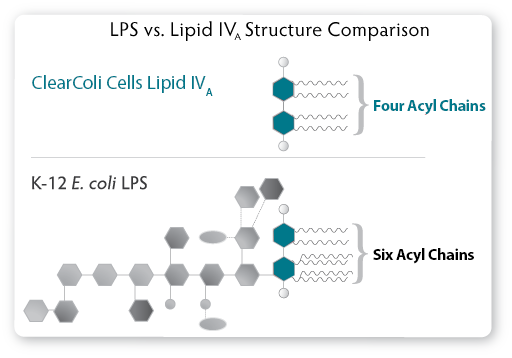 |
| Figure 1. The LPS of a normal E. coli cell compared to the genetically modified Lipid IVA from ClearColi cells.
In ClearColi, the oligosaccharide chain has been deleted, and two of
the six acyl chains have been removed to disable the endotoxin signal. |
Modifications to the genotype of the ClearColi cells consist of seven
separate gene deletions, thereby ensuring there is no chance of genetic
reversion back to wild type and production of normal LPS. These
mutations result in the deletion of the oligosaccharide chain from the
LPS, making it easier to remove the resulting lipid IVA from
the downstream product. More importantly, two of the six acyl chains are
deleted. The six acyl chains of the LPS are the trigger which is
recognised by the Toll-like receptor 4 (TLR4) in complex with myeloid
differentiation factor 2 (MD-2), causing activation of NF-?B and
production of proinflammatory cytokines. Lipid IVA, which
contains only four acyl chains, is not recognised by TLR4 and thus does
not trigger the endotoxic response (see Figure. 2).
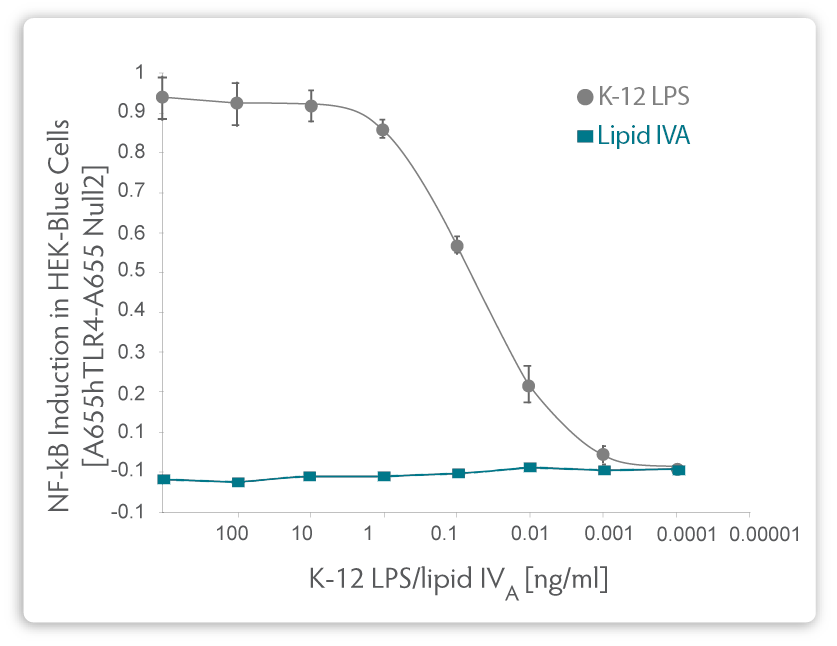 |
| Fig. 2. Comparison of relative NF-κB induction in HEK-Blue Cells using purified LPS from a K-12 E. coli strain or from pure, synthetically manufactured Lipid IVA. |
Back to top ?
ClearColi BL21(DE3) competent cells have the following genotype:
F– ompT hsdSB (rB- mB-) gal dcm lon λ(DE3 [lacI lacUV5-T7 gene 1 ind1 sam7 nin5]) msbA148 ΔgutQ ΔkdsD ΔlpxLΔlpxMΔpagPΔlpxP ΔeptA
Seven specific deletion mutations (ΔgutQ ΔkdsD ΔlpxL ΔlpxMΔpagPΔlpxPΔeptA) encode the modification of LPS to Lipid IVA, while one additional compensating mutation (msbA148) enables the cells to maintain viability in the presence of the LPS precursor lipid IVA.
Back to top ?
Why Endotoxin Removal is not the Best Method
Lipopolysaccharide (LPS), also known as endotoxin, is a potent agonist
for hTLR4/MD-2-mediated proinflammatory activity in human immune cells.
To prevent toxicity, recombinant proteins, commonly manufactured in E. coli,
must be essentially free of endotoxin. However, efficient elimination
of endotoxin is a challenging task, and none of the downstream
applications remove endotoxin entirely. Current methods for endotoxin
removal from recombinant proteins are varied, including
ultra-filtration, activated carbon, surfactants, anion exchange
chromatography, and immobilized sepharose. Each of these strategies
involves negative effects: significant yield loss, high cost, loss of
bioactivity of the protein, or bioactivity of the additives used for
endotoxin clean up. Table. 1 below describes some of the most common
methods and their associated shortcomings:
| Table 1: |
|
| Method | Disadvantages |
| Ultrafiltration | - Only useful for small proteins
- Endotoxin monomers may permeate the membrane due to endotoxin/protein interaction
- Inefficient for proteins which can be damaged by physical forces
|
| Activated carbon | - Adsorbing activity for both endotoxin and protein
|
| Surfactants | - Expensive
- May affect bioactivity of protein
- Difficult to completely remove
- Removal may lead to product loss
|
| Anion-exchange chromatography | - High adsorbtion of both endotoxin and acidic protein
- No selectivity to adsorb endotoxin
|
| Histamine- and histidine-immobilized Sepharose | - Removing capacity dependent on the ionic strength
- Biological activity of histamine
|
| Polymyxin B-immobilised Sepharose | - Protein losses due to the ionic interaction between polymyxin B and protein
- Polymixin B is physiologically active
|
Back to top ?
Protein Expression with ClearColi
The relative production efficiency of endotoxin free ClearColi™
BL21(DE3) competent cells was compared to normal BL21(DE3) cells using
two different recombinant proteins. Although overall growth rates of the
ClearColi are slower, final protein production levels are very similar
(see Figure. 3 and 4).
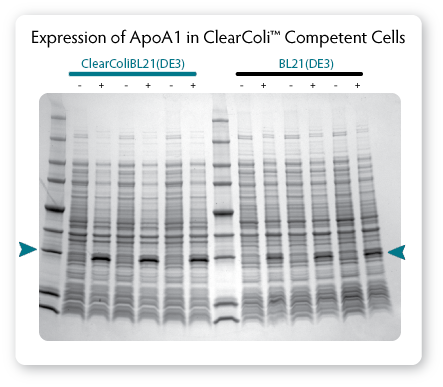 |
| Figure 3. Comparison of protein expression in ClearColi BL21(DE3) and Lucigen's E. Cloni® EXPRESS BL21(DE3) competent cells.
Cells containing a T7 expression plasmid harbouring a gene encoding the
human apolipoprotein A1 (ApoA1) were grown in LB Miller medium at 37
°C. When cultures reached OD600 of 0.6 to 0.8, expression was
induced by the addition of 0.4 mM IPTG and incubation was continued for
3 hours. Equivalent numbers of uninduced (-) and induced ( ) cells were
lysed by heating in Laemmli buffer and samples were analysed by
SDS-PAGE on a 4% - 20% polyacrylamide gradient gel. |
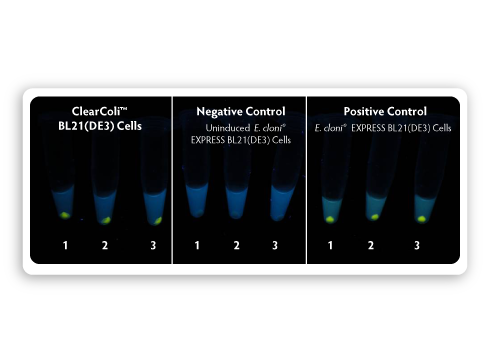 |
| Figure 4. Comparison of fluorescent protein expression in ClearColi BL21(DE3) and E. cloni EXPRESS BL21(DE3) cells.
A pET expression vector harbouring a gene encoding a yellow fluorescent
protein (LucY) under the control of a T7 promoter was transformed into
both ClearColi BL21(DE3) and E. cloni EXPRESS BL21(DE3). Colonies were
inoculated into LB-Miller medium and grown at 37 °C for induction.
Samples of induced and uninduced cells containing equivalent numbers of
cells were centrifuged, and cell pellets were photographed under
long-wavelength UV illumination. |
Back to top ?
Endotoxicity Assay Results
In most cases, a simple nickel column purification is sufficient for
endotoxic shock response negative recombinant proteins suitable or
downstream eukaryotic cellular response assays. Researchers can now
assay target protein effects without concern that immune response
triggers are a result of LPS endotoxin contamination. To demonstrate,
ApoA1 protein expressed from ClearColi BL21(DE3) competent cells was
Ni-column purified and tested for endotoxicity using both the HEK-Blue
cell line from Invivogen (see Fig 5) and the LAL assay (see Fig. 6).
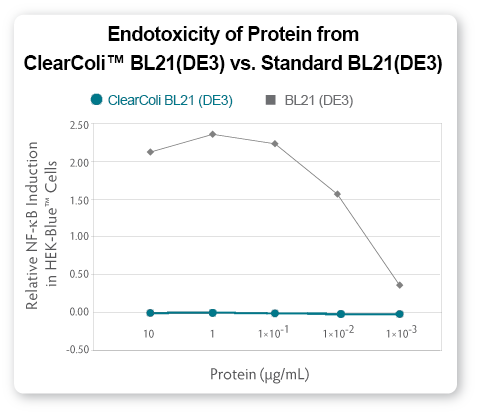 |
| Figure
5. Comparison of endotoxic response from protein derived from ClearColi
BL21(DE3) and traditional BL21(DE3) competent cells |
Back to top ?
LAL Testing: Does it really measure endotoxin?
Limulus
amebocyte assay testing is an FDA-approved method for detection of
endotoxins and the most common assay used; however the LAL assay is
activated solely by the 4´-monophosphoryldiglucosamine backbone of LPS.
LAL activity is minimally influenced by acylation pattern of LPS, the
key determinant of endotoxicity in eukaryotic cells. The LAL assay also
recognizes a wider spectrum of LPS/lipid A variants than the central
cellular endotoxin sensor system of the human immune cell system. As
such, false positive results can and will result due to the lack of
specificity of the assay.
A simple Ni-column purification step
for proteins produced from ClearColi cells will reduce LAL response
levels by 95% or greater (see Fig. 6). However, the residual EU
measurements are due to the non-specific nature of the assay unless
extraneous LPS contamination from other sources is present. Alternative
toxicity assays, such as those using HEK-Blue cells (see Fig. 5) suggest
that even in the presence of EU levels above threshholds normally
targeted by researchers, the actual immunogenic effects from
ClearColi-derived proteins are non-existent.
Due to the non-specificity of the LAL assay when combined with lipid IVA from ClearColi, it is suggested that researchers consider alternative methods of endotoxin measurement.
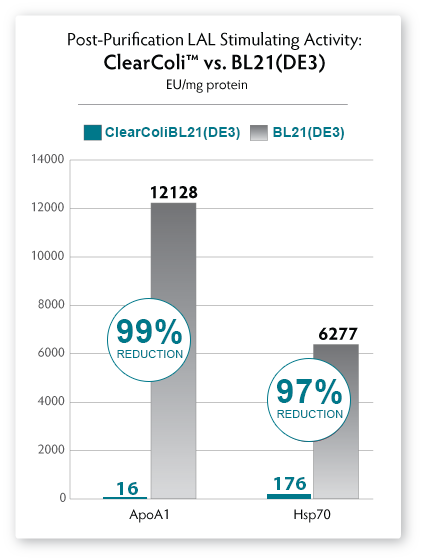 |
| Figure
6. Comparison of endotoxin units as measured by the LAL assay using
nickel-column purified recombinant ApoA1 and HSP protein expressed in
ClearColi BL21(DE3) (red bars) and E. cloni EXPRESS BL21(DE3) (grey
bars) competent cells. Protein expressed from ClearColi demonstrates significant reduction in EU/mg without endotoxin removal steps. |
Back to top ?
Growth Rates for ClearColi BL21(DE3) Cells
ClearColi BL21(DE3) cells grow at approximately 50% of the rate of
normal BL21(DE3) cells (see Fig. 7). Users should expect to see very
small colonies for the first 24 hours after plating transformants. We
recommend incubating plates for 32-40 hours before picking colonies for
future experiments. When grown to sufficient densities, ClearColi
BL21(DE3) cells produce similar protein levels as normal BL21(DE3) cells
when comparing equal numbers of cells.
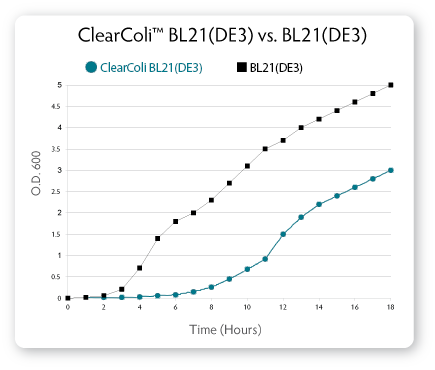 |
| Figure 7. Comparison of growth rates for ClearColi BL21(DE3) vs. E. cloni EXPRESS BL21(DE3) cells. Cells were inoculated to an initial OD600 of ~0.003 in 200 ml of LB Miller medium and grown at 37° C with shaking at 210 rpm. The OD600 of the cultures was recorded hourly. |
Back to top ?
Relevant Reference Articles:
- Teghanemt, et al, Molecular Basis of Reduced Potency of Underacylated Endotoxins, J Immunol, 2005; 175:4669-4676
-
Mamat, et al, Single amino acid substitutions in either YhjD or MsbA
confer viability to 3-deoxy-D-manno-oct-2-ulosonic acid-depleted Escherichia coli, Molecular Microbiology, 2008, 67(3), 633–648
- Meredith, et al, Redefining the Requisite Lipopolysaccharide Structure in Escherichia coli, ACS Chemical Biology, 2006, 1(1), 33-42
-
Brandenburg, et al, The Expression of Endotoxic Activity in the Limulus
Test as Compared to Cytokine Production in Immune Cells, Current Medicinal Chemistry, 2009, 16, 2653-2660
-
Gutsmann, et al. Structural prerequisites for endotoxic activity in the
Limulus test as compared to cytokine production in mononuclear cells, Innate Immunity, 2010, 16(1), 39-47
- Beom Seok Park1 et al., The structural basis of lipopolysaccharide recognition by the TLR4–MD-2 complex, Nature 458, 1191-1195 (30 April 2009)
Back to top ?
ClearColi Licensing Information:
ClearColi™ Competent cells are subject to US Patent 8,303,964 and other US and foreign pending patents.
Lucigen Corporation ("Lucigen") has a license from Research Corporation Technologies
to sell ClearColi competent cells to third-parties for non-commercial
research purposes only. A separate license is required for any
commercial use. For more information about the use of this product by
commercial entities, please review our full licensing page.
Back to top ?
Transformation efficiency:
1 × 109 cfu/ug, measured using pUC19 DNA in standard electroporation conditions
Genotype:
F– ompT hsdSB (rB- mB-) gal dcm lon λ(DE3 [lacI lacUV5-T7 gene 1 ind1 sam7 nin5]) msbA148 ΔgutQΔkdsD ΔlpxLΔlpxMΔpagPΔlpxPΔeptA
SDS
Manuals and user guides
Product information sheets
FAQs
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200