Cryophile Proteinase (heat-labile)
Histones and other proteins are known to protect nucleic acids from interacting optimally with other DNA binding proteins and enzymes. Cryophile Proteinase (heat inactivated) is ideally suited for transforming chromatin and other dense protein-bound nucleic acids to their naked form. The enzyme is very easily heat-inactivated thus allowing thermal inactivation at temperatures enabling RNA integrity as well as avoiding dissociation of dsDNA.
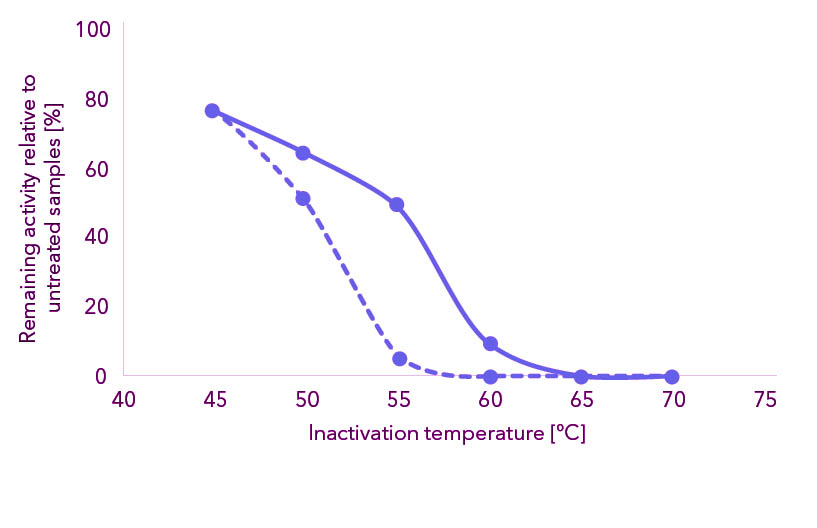
Figure 1: Cryophile Proteinase is easy to inactivate after use. Figure shows remaining activity after incubation of Cryophile Proteinase at different temperatures. Incubation for 30 minutes at indicated temperature in presence of 50 (solid line) or 300 (dotted line) mM NaCl. Remaining activity in % of sample kept on ice in same buffer.
Unit definition
One unit is defined as the amount of enzyme that produces one μmol 4-nitroaniline (extinction coefficient 8.8 mM-1 at 410 nm) at 25°C, pH 8.0 per minute (buffer conditions 50 mM Tris-HCl pH 8.0, 1% DMSO, 1 mM Suc-Ala-Ala-Pro-Phe-pNA).
Optimal reaction conditions
Optimal enzyme activity is between pH 7–10. Recommended temperature range for activity is from 25–40°C. Cryophile Proteinase is not dependent of divalent cations such as calcium (Ca2 ) for activity, and is therefore active in buffers containing EDTA (<40°C reaction temperature). Cryophile Proteinase is compatible with digestion of proteins in the presence of SDS (0.2–1%) and urea (1–5 M). The enzyme tolerates 500 mM guanidine thiocyanate and 1% Triton X-100 (>50% activity). No significant loss of activity at 400 mM NaCl is observed.
Inactivation
Recommended inactivation conditions are incubating the enzyme at 60°C for 15–30 min. Inactivation temperature and time can be adjusted to suit desired application or stringency on residual protease activity. Cryophile Proteinase is susceptible to EDTA and DTT at elevated temperatures (>40°C), so EDTA or DTT will give additive effect upon inactivation.
Inhibitors
The enzyme is inhibited by general serine protease inhibitors, such as PMSF.
Storage and stability
Optimal stability and storage condition is between pH 6–9. The enzyme is stable upon storage at -20°C for >1 year. Cryophile Proteinase is also stable upon storage at 4°C (>6 months) and room temperature (>4 weeks). Cryophile Proteinase is stable at dilute concentrations (<0.1 mg/ml) in solutions.
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200