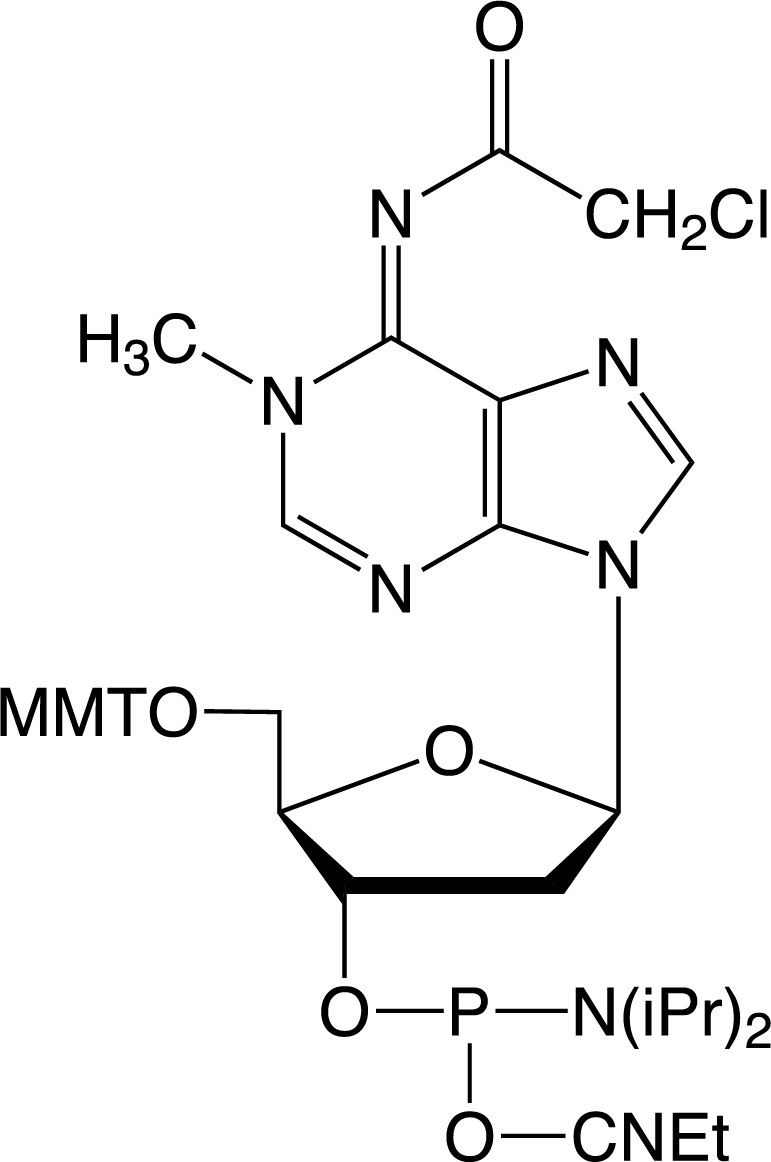
| 1-Me-dA-CE Phosphoramidite |
Catalog Number: 10-1501-xx
Description: 1-Me-dA-CE Phosphoramidite
5'-Monomethoxytrityl-N6-chloroacetyl-1-methyl-2'-deoxyAdenosine,
3'-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite |
| Formula: C42H49ClN7O6P |
M.W.: 814.31 |
F.W.: 328.24 |
Diluent: Anhydrous Acetonitrile |
| Coupling: No changes needed from standard method recommended by synthesizer manufacturer. Monomers that allow for UltraMILD deprotection are recommended. (dA:10-1601-xx, dC: 10-1015-xx, dG: 10-1621-xx, dT: 10-1030-xx ). To avoid any exchange of the iPr-Pac group on the dG with acetyl, use the UltraMild Cap Mix A (40-4210-xx/ 40-4212-xx). 0.02M Iodine for oxidation is also recommended. |
| Deprotection: 2 M Ammonia in methanol, 24 hours at Room Temperature using UltraMILD amidites or 60 hours at Room Temperature if using standard amidites. |
| Storage: Refrigerated storage, maximum of 2-8°C, dry |
| Stability in Solution: Similar to dA,C,G,T-CE Phosphoramidites |
MUTAGENESIS
Cellular polynucleotides are alkylated by endogenous components, such as S-adenosylmethionine, or after reacting with two general classes of environmental and laboratory chemicals. SN1 chemical agents include alkylnitrosourea and N-alkyl-N-nitro-N-nitrosoguanidine that react with the N7 position of guanine, N3 of adenine, O6 of guanine, O2 or O4 of pyrimidines, and the non-phosphodiester oxygen atoms of the phosphate backbone. In contrast, SN2 chemical agents such as methyl methanesulfonate and dimethyl sulfate react primarily with the N1 position of adenine (1-Methyl-2’-deoxyadenosine) and N3 of cytosine. To avoid chain branching during synthesis when using DCI as activator, N6-Me-dA is offered with acetyl protection.
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200